AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Adobe zii 2021 6.0.512/3/2023 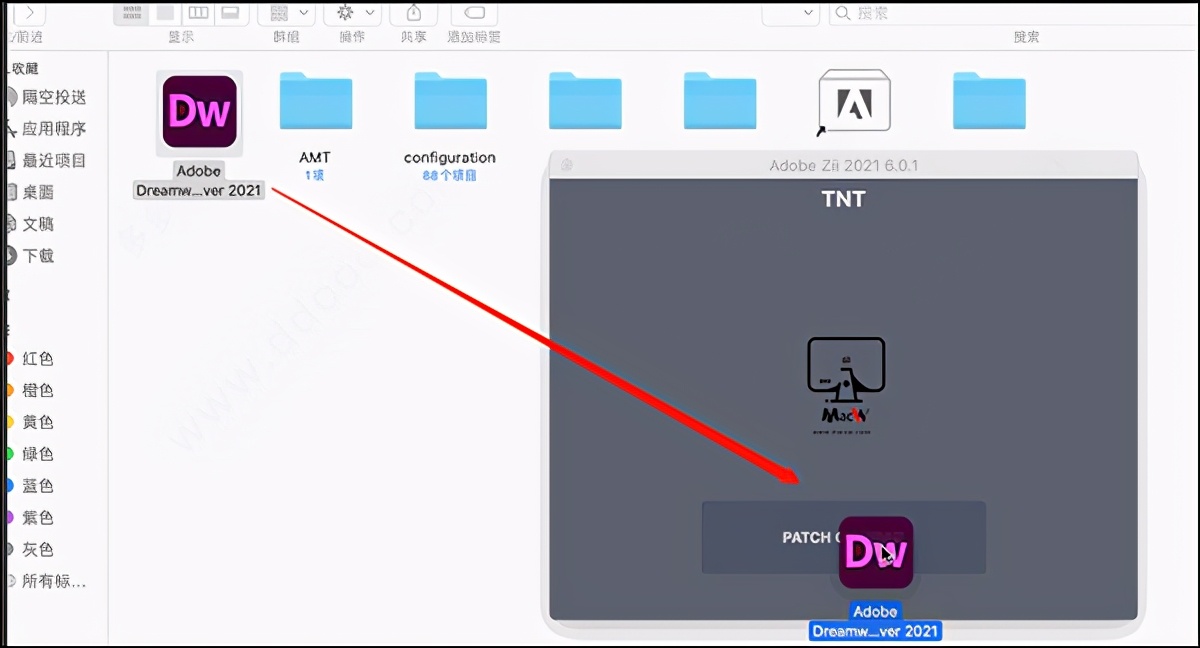
How to Install Adobe Patcher Zii (Guide): Do click on any place and just close the window. New version adobe zii patcher 6.1.6 coming soonĪfter testing and cracking LR and AU are completed, you will have the LR and AU popped up on a window. 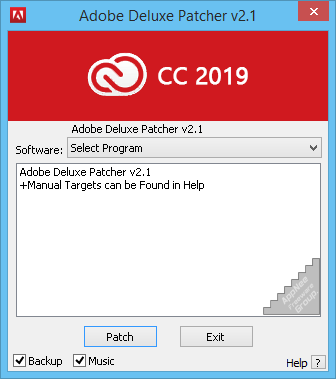
OR Check Our Release Note/NewsClick here Supported Products: Adobe Zii Download Process Tutorial Link Need – Click Here If First Link Not Working your Mac try our Different Adobe Zii 6.1.5 Below Link Some of The New supports By Adobe Zii Patcher For Mac: What’s New or Fixed? 
Download now also save our website your browser bookmark area. Here you find adobe Adobe zii for mac direct link, so you now visits right place because here our AdobeZii.Com share below one click direct link also our link safe and virus free. Adobe Zii 2021 is a small and great utility which helps you forget about all issues with the registration of all Adobe existing products. As of March 2021, Photoshop now runs natively on Apple computers using the Apple Silicon M1 chip with 1.5X the speed of similarly configured previous generation systems. Run Photoshop faster in native mode on Apple computers using the Apple Silicon M1 chip. Its capabilities very easily taking over for every area.
0 Comments
Read More
Back to Blog
Chromium table of elements12/3/2023 
For example, in the equation Fe 2+ + 2 e − ⇌ Fe( s) (–0.44 V), the Gibbs energy required to create one neutral atom of Fe( s) from one Fe 2+ ion and two electrons is 2 × 0.44 eV = 0.88 eV, or 84 895 J/mol of electrons, which is just the Gibbs energy of formation of an Fe 2+ ion, since the energies of formation of e − and Fe( s) are both zero. After dividing by the number of electrons, the standard potential E° is related to the standard Gibbs free energy of formation ΔG f° by:Į = ∑ Δ G left − ∑ Δ G right F where F is the Faraday constant.For example, the equation Fe 2+ + 2 e − ⇌ Fe( s) (–0.44 V) means that it requires 2 × 0.44 eV = 0.88 eV of energy to be absorbed (hence the minus sign) in order to create one neutral atom of Fe( s) from one Fe 2+ ion and two electrons, or 0.44 eV per electron, which is 0.44 J/C of electrons, which is 0.44 V. All of the reactions should be divided by the stoichiometric coefficient for the electron to get the corresponding corrected reaction equation. Although many of the half cells are written for multiple-electron transfers, the tabulated potentials are for a single-electron transfer.The relation in electrode potential of metals in saltwater (as electrolyte) is given in the galvanic series. An activity of unity for each pure solid, pure liquid, or for water (solvent).This pressure is used because most literature data are still given for this value (1 atm) rather than for the current standard of 100 kPa (1 bar) presently considered in the standard state. A partial pressure of 101.325 kPa (absolute) (1 atm, 1.01325 bar) for each gaseous reagent.An effective concentration of 1 mol/L for each aqueous species or a species in mercury amalgam (an alloy of mercury with another metal).The data values of standard electrode potentials ( E°) are given in the table below, in volts relative to the standard hydrogen electrode, and are for the following conditions: Main article: Standard electrode potential |
 RSS Feed
RSS Feed
